Rates of reaction презентация
Содержание
- 3. What does rate of reaction mean?
- 4. Rates of reaction
- 5. Reactions, particles and collisions
- 6. Changing the rate of reactions
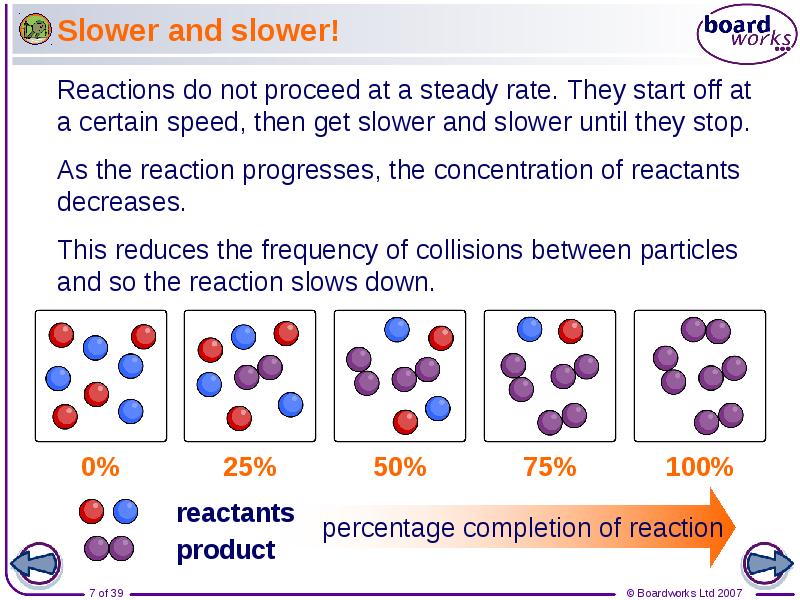
- 7. Slower and slower!
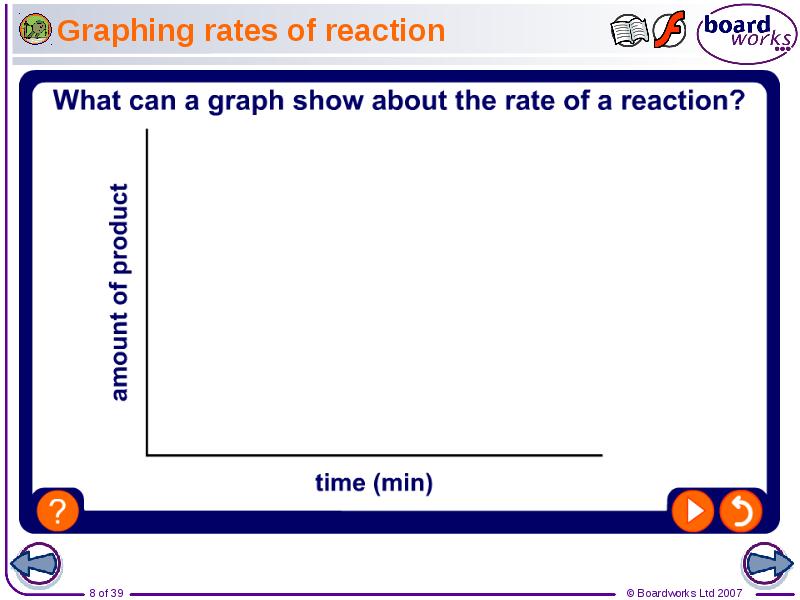
- 8. Graphing rates of reaction
- 9. Reactant–product mix
- 10. How can rate of reaction be measured?
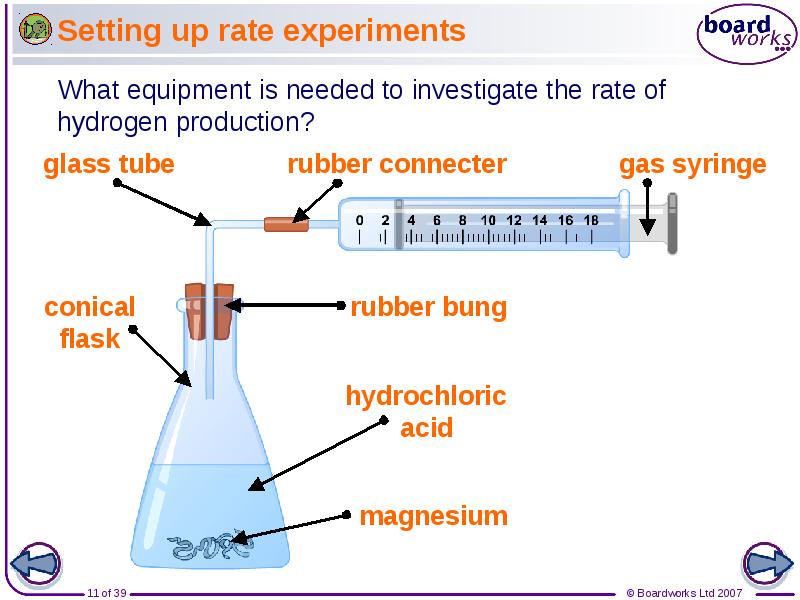
- 11. Setting up rate experiments
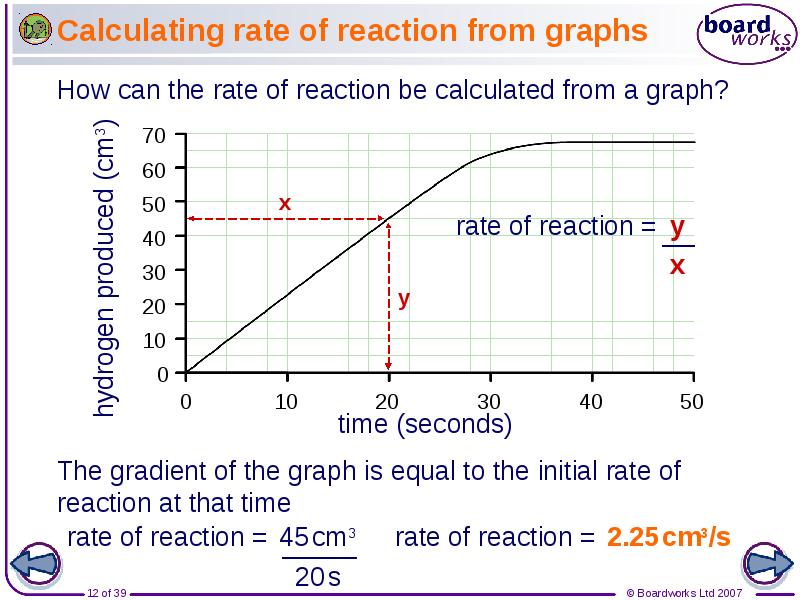
- 12. Calculating rate of reaction from graphs
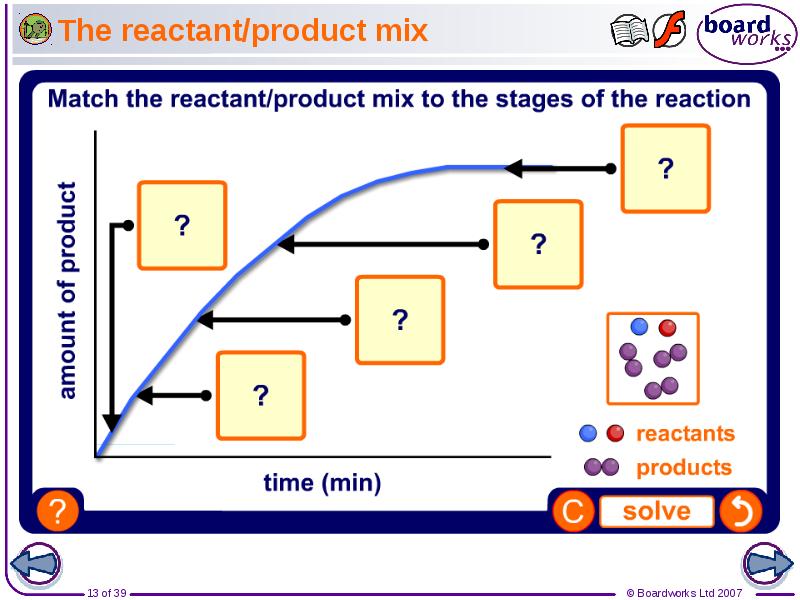
- 13. The reactant/product mix
- 14. Collisions and reactions: summary
- 16. Temperature and collisions
- 17. Effect of temperature on rate
- 18. Temperature and particle collisions
- 19. Temperature and batteries
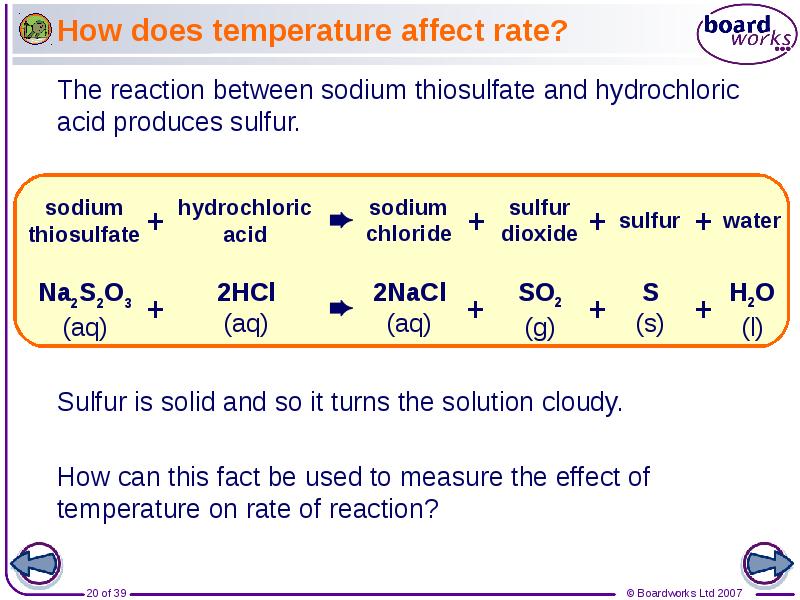
- 20. How does temperature affect rate?
- 21. The effect of temperature on rate
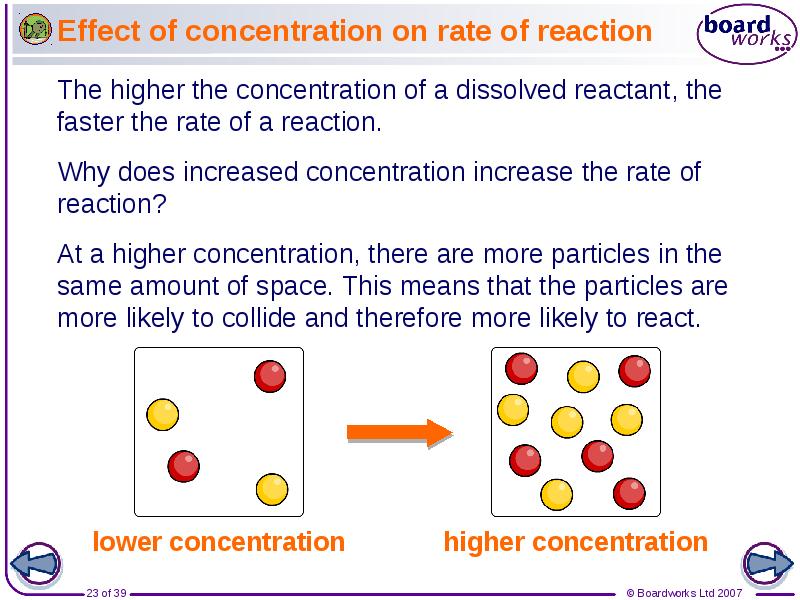
- 23. Effect of concentration on rate of reaction
- 24. Concentration and particle collisions
- 25. The effect of concentration on rate
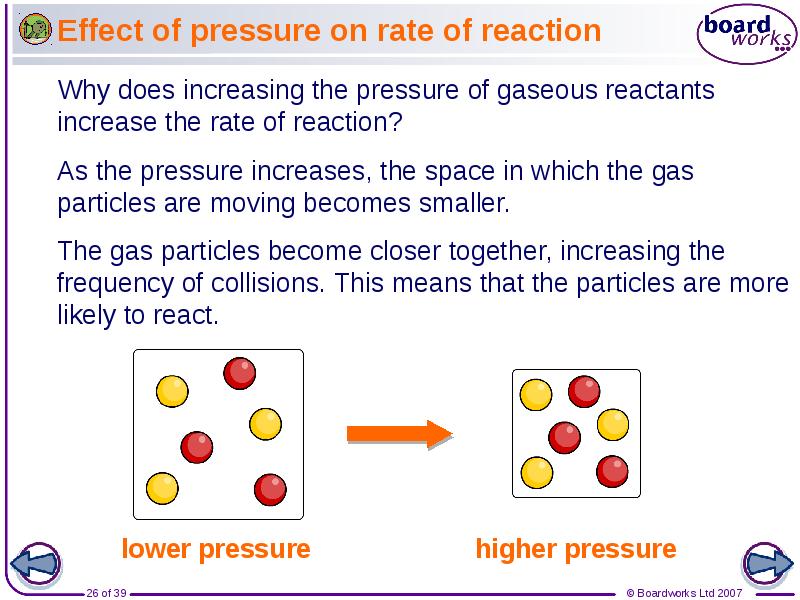
- 26. Effect of pressure on rate of reaction
- 28. Effect of surface area on rate of reaction
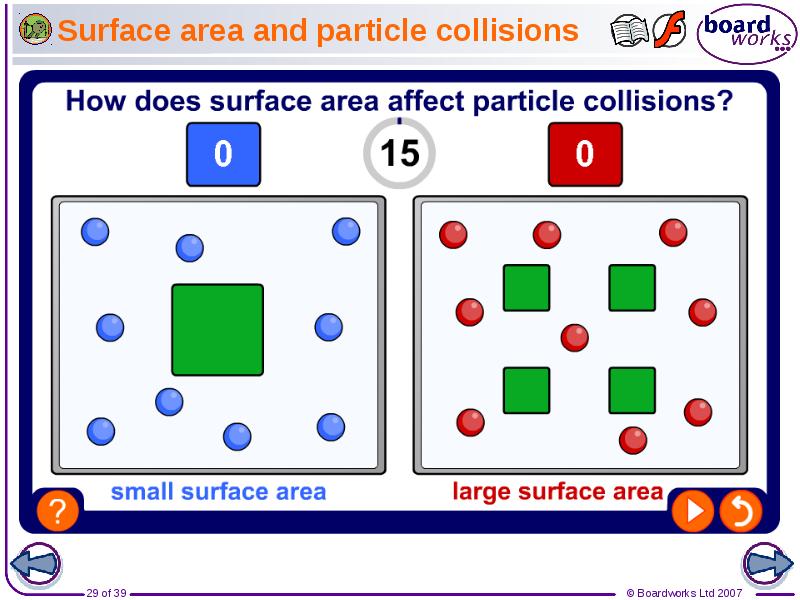
- 29. Surface area and particle collisions
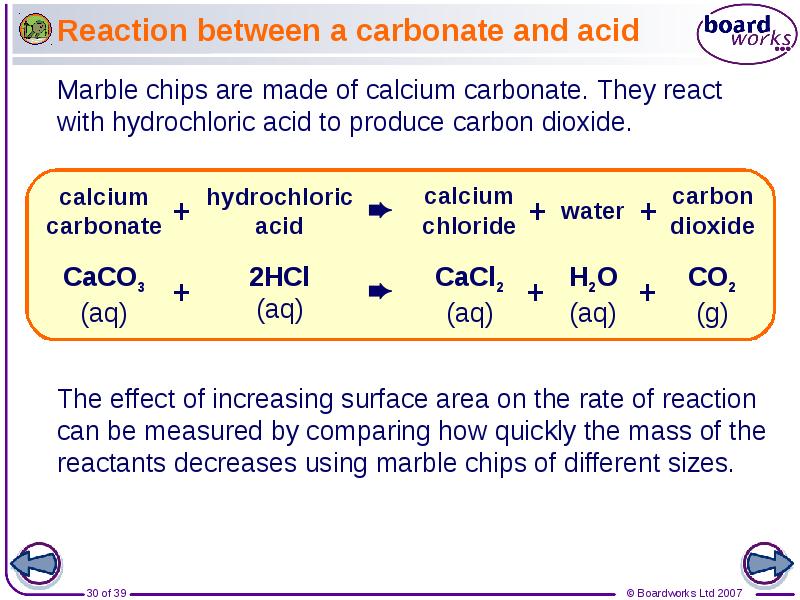
- 30. Reaction between a carbonate and acid
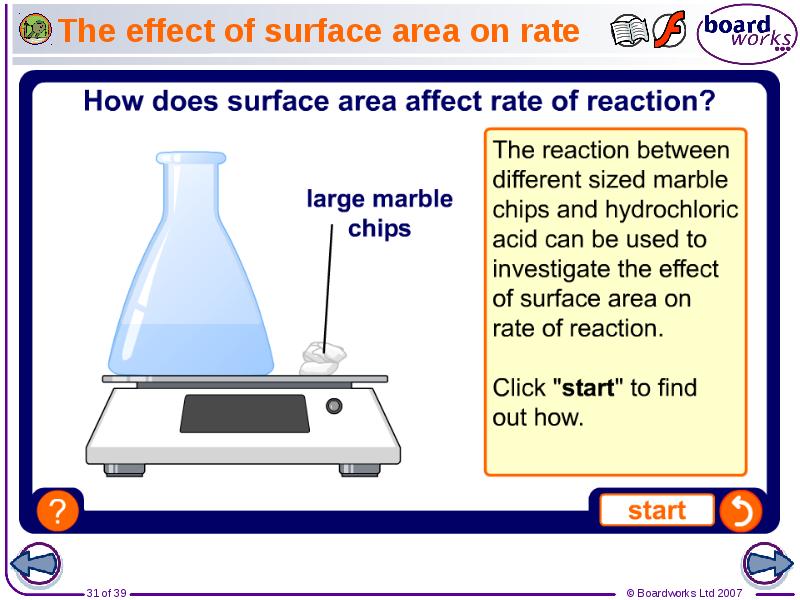
- 31. The effect of surface area on rate
- 32. What are catalysts?
- 33. Everyday catalysts
- 34. Catalysts in industry
- 36. Glossary
- 37. Anagrams
- 38. Rates of reaction: summary
- 39. Multiple-choice quiz
- 40. Скачать презентацию























Слайды и текст этой презентации
Похожие презентации